Inside the Clinical Diagnostics Industry – Extract 2
Chapter 12 — Evolution of Laboratory Systems Integration Burden
The following chapter extract is taken from Chapter 12 of Inside the Clinical Diagnostics Industry – Constraints Shaping Strategy – Towards Health Intelligence. It addresses the structural consequences of integration.
Analytical comparability required coordination. As instruments standardised measurement, laboratories integrated workflows to sustain throughput and reliability.
Middleware connected platforms. Automation orchestrated processes. Multi-vendor environments required harmonisation. Integration reduced manual discretion but increased systemic dependency.
Each interface introduced coordination requirements. Instrument-level integration expanded into workflow orchestration and cross-domain alignment. The laboratory became a coordinated subsystem rather than an isolated analytical unit.
Integration improved performance. It also increased coupling. Local optimisation became constrained by global configuration. Independent evolution became more difficult once interoperability was embedded.
The integration burden intensified as laboratories consolidated and healthcare systems centralised. Diagnostics functioned within broader institutional architectures.
Integration did not replace analytical discipline. It layered coordination on top of it. Structural exposure increased accordingly.
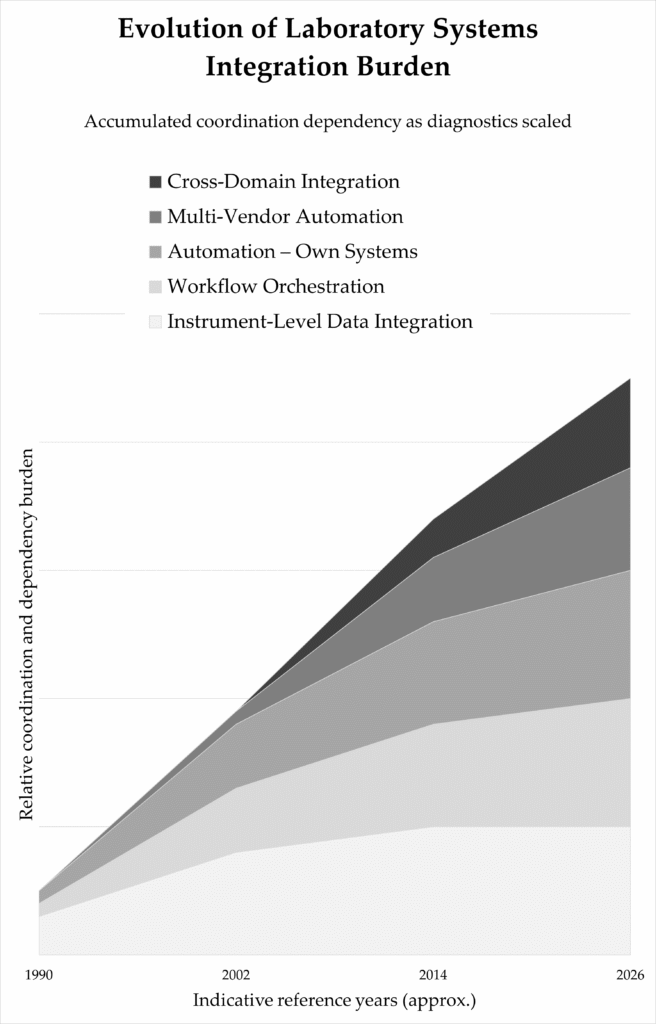
Indicative growth of coordination and integration requirements across analytical, operational, and cross-domain environments (1990–2026).
