Inside the Clinical Diagnostics Industry – Extract 1
Prelude — Cumulative Deployment Requirements in Clinical Diagnostics
The following chapter extract is taken from the Prelude of Inside the Clinical Diagnostics Industry – Constraints Shaping Strategy – Towards Health Intelligence. It introduces the structural accumulation of deployment requirements.
The evolution of clinical diagnostics cannot be understood as a succession of technological replacements. Each phase of development introduced new requirements without eliminating the previous ones.
Analytical systematisation established reproducibility and trust. Quality systems formalised reliability. Platforms became scalable.
Systems integration added coordination. Instruments were connected. Data flowed across environments. Automation reduced variability but increased configuration dependency.
Specialisation introduced depth. Diagnostics aligned more closely with specific diseases and therapeutic pathways. Interpretation became context-sensitive.
Interdependence followed. Regulation intensified. Evidence requirements expanded. Deployment became inseparable from healthcare governance structures.
These layers accumulated. Analytical reliability did not dissolve when integration expanded. Integration did not disappear when specialisation intensified. Each requirement remained active.
Strategic decisions now operate across all layers simultaneously. Deployment demands analytical robustness, operational coordination, interpretive alignment, and regulatory conformity.
The industry did not become complex through failure. It became complex through cumulative success.
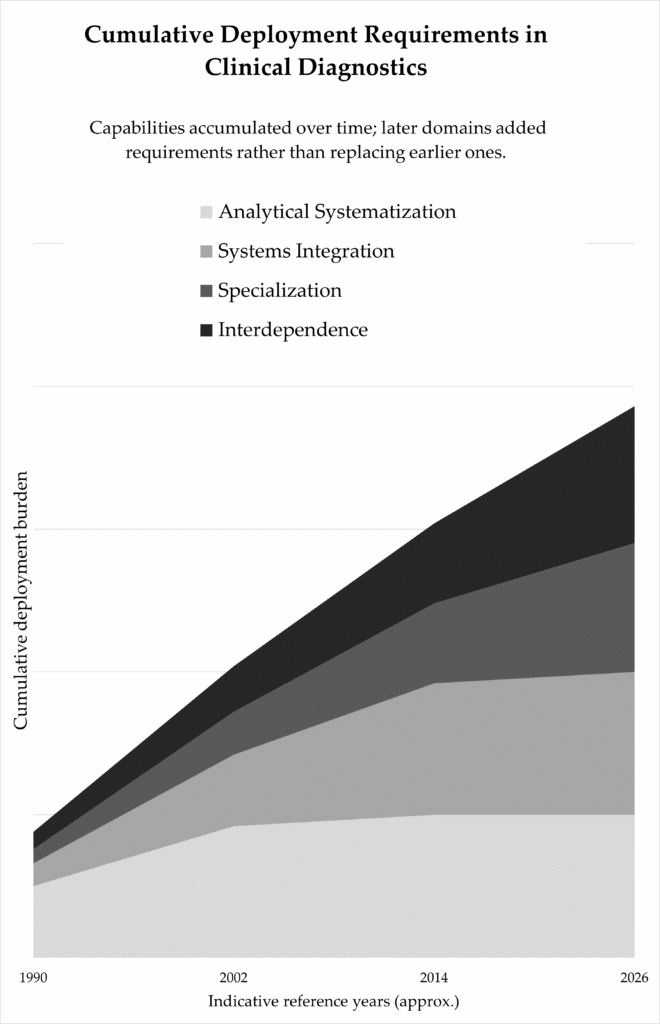
Indicative accumulation of structural requirements across analytical systematisation, systems integration, specialisation, and interdependence (1990–2026).
